_This web page was produced as an assignment for Genetics 677, an undergraduate course at UW-Madison.
Background
Images taken from STRING database.
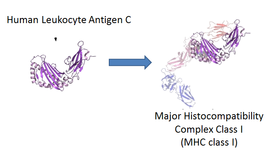
Psoriasis is an autoimmune disease affecting the skin in over 1.5 million Americans. It causes red, scaly patches of skin, called plaques, to occur all over the body, mostly on the knees, elbows, lower back, and scalp. Plaques tend to flare up from environmental factors like stress, infection, and injuries. Studies have linked the disease to many genes, most commonly to HLA-C. HLA-C, or Human Leukocyte Antigen C, is a protein that functions together with the Major Histocompatibility Complex Class I (MHC class I). HLA-C/MHC class 1 functions as a part of the immune system to present antigens (degraded protein fragments) to T-cells (type of white blood cell) to activate an immune response in the epidermis (skin layer). After T-cells receive the antigens, an immune reaction occurs to release cytokines which induces a cell proliferation response. This increase in cell proliferation is responsible for the psoriasis plaque formation.
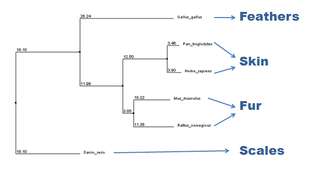
Question 1: Why doesn't psoriasis occur in feathered or scaled organisms?
Upon looking at the phylogeny of the HLA-C, many "skin" types were represented. So HLA-C was observed in all sorts of skin types: skin, fur, feathers, and scales. Looking further, only two skin types are known to develop psoriasis, that being skin and fur (which brings me to question one). Since HLA-C is the predominant locus responsible for psoriasis, what is different about HLA-C in feathers and scaled skin types that makes it impossible to obtain psoriasis? To answer this questions, the relationship of HLA-C in various skin types must be further investigated.
Model Organism: Zebrafish
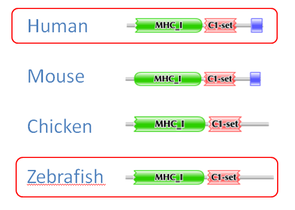
Domains were identified for homologs using Pfam.
Looking at the HLA-C homolog domains, they are fairly consistent. Initially, the blue domain initially seems like a likely candidate psoriasis development; however, this region consists of the C-terminus cytoplasmic tail. This region has been noted to have no role in protein functionality. Overall, the domain comparisons suggest that the HLA-C structure/sequence is not the reason for psoriasis development.
Taking advantage of the domain consistency, the zebrafish would serve as a good model for further investigation. The zebrafish serves as a good model because is the most distant ancestor, has no psoriasis occurrence, and has many genetic tools widely available.
Taking advantage of the domain consistency, the zebrafish would serve as a good model for further investigation. The zebrafish serves as a good model because is the most distant ancestor, has no psoriasis occurrence, and has many genetic tools widely available.
Question 2: Does HLA-C function the same in zebrafish as it does in humans?
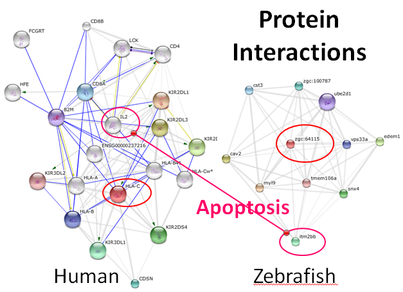
Protein interaction networks were identified using STRING database.
The human HLA-C interactome (protein interaction network) was compared to the zebrafish HLA-C homolog for similarities. The two interactomes shared a common function, apoptosis, which was identified using the GO Term Finder. Apoptosis was most significantly carried out by IL2 in humans and itm2bb in zebrafish. As a reminder, psoriasis symptoms arise from an over production of skin cells, caused from an overactive immune response. This highlights the significance of a functional apoptosis pathway.
In humans, IL2 (Interleukin 2) is a cytokine released in T-cells when HLA-C/MHC class 1 presents antigens. When IL2 is released a immune response is triggered creating an increase in cell proliferation. When IL2 is not released another protein, itm2bb (intergral membrane protein 2bb), is expressed to induce apoptosis in T-cells. Zebrafish contain a functional IL2 homolog; so, recognizing the human relationship, how does IL2 and itm2bb function independently and together in zebrafish?
Due to the conserved domains of IL2 and itm2bb, the function between both proteins should mirror the human relationship. Investigating this relationship will better our understanding of Zebrafish HLA-C interactions in order to understand why ‘scaled’ organisms don’t get psoriasis. Being able to identify the differences between the two organisms could provide clues to the underlying reason for developing psoriasis.
In humans, IL2 (Interleukin 2) is a cytokine released in T-cells when HLA-C/MHC class 1 presents antigens. When IL2 is released a immune response is triggered creating an increase in cell proliferation. When IL2 is not released another protein, itm2bb (intergral membrane protein 2bb), is expressed to induce apoptosis in T-cells. Zebrafish contain a functional IL2 homolog; so, recognizing the human relationship, how does IL2 and itm2bb function independently and together in zebrafish?
Due to the conserved domains of IL2 and itm2bb, the function between both proteins should mirror the human relationship. Investigating this relationship will better our understanding of Zebrafish HLA-C interactions in order to understand why ‘scaled’ organisms don’t get psoriasis. Being able to identify the differences between the two organisms could provide clues to the underlying reason for developing psoriasis.
Experimental Design
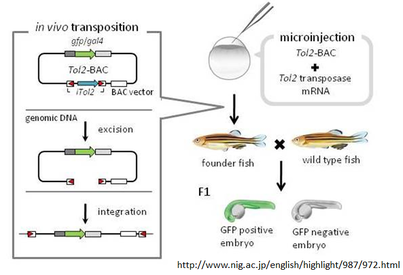
To investigate the the function of IL2 and itm2bb in zebrafish, two transgenic lines will be created, a IL2 knockout and an itm2bb knockout. Creating a transgenic knockout, a series of steps must undergo. First, the gene of interest (using restriction enzymes) must be inserted into a plasmid containing glow factor protein (GFP) and the BAC (knockout) vector. Second, microinjection will insert plasmid to allow for genome integration. Finally, after undergoing homologous recombination the gene of interest will no longer function, creating a transgenic knockout. This will be identifiable easily because GFP a part of the integrated vector.
Vitamin D3
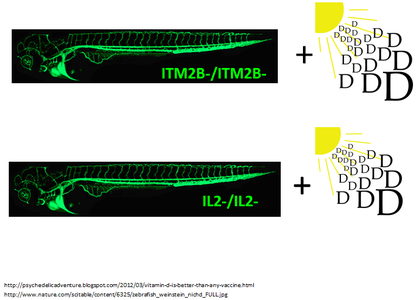
Vitamin D3 has been shown to suppress IL2 expression, thus decreasing cell proliferation and reducing psoriatic plaques. Does Vitamin D3 affect IL2 expression in zebrafish too? To answer this question, the IL2 and itm2bb knockout transgenic zebrafish will undergo vitamin D3 exposure by introducing vitamin D3 into their environment.
Microarray Experiment

HLA-C expression/interactions differences between skin and scales gives insight on the cause of psoriasis.
A microarray experiment will be conducted to measure the protein expression levels in zebrafish (see left figure). These expression levels will be normalized by also measuring the wildtype levels. The results should answer the following questions:
Does zebrafish IL2 and itm2bb have the same expression relationship as it does in humans? (itm2bb expression levels increase when IL2 expression is low)
Does Vitamin D3 affect IL2 expressions like in humans? (IL2 expression is inhibited by vitamin D3 in humans)
A microarray experiment will be conducted to measure the protein expression levels in zebrafish (see left figure). These expression levels will be normalized by also measuring the wildtype levels. The results should answer the following questions:
Does zebrafish IL2 and itm2bb have the same expression relationship as it does in humans? (itm2bb expression levels increase when IL2 expression is low)
Does Vitamin D3 affect IL2 expressions like in humans? (IL2 expression is inhibited by vitamin D3 in humans)
Expected Results
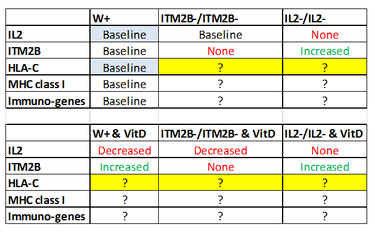
IL2 knockouts should result in no IL2 expression and increased itm2bb expression.
Itm2bb knockouts should result in no itm2bb expression.
IL2 knockouts + Vitamin D3 should still result in increased itm2bb expression.
Itm2bb knockouts + Vitamin D3 should result in decreased IL2 expression.
Wildtype should establish baseline expression levels.
Wildtype + Vitamin D should result in decreased IL2 levels, thus increased itm2bb levels.
Other immune system gene expression levels will be measured (such as other cytokines, growth factors, MHC class 1 genes) if their homolog exists. This will provide immense insight into understanding how the zebrafish immune system works. The zebrafish immune system can be compared to the human immune system so that differences can be established. These differences can be studied to see why scaled organisms don't get psoriasis.
Itm2bb knockouts should result in no itm2bb expression.
IL2 knockouts + Vitamin D3 should still result in increased itm2bb expression.
Itm2bb knockouts + Vitamin D3 should result in decreased IL2 expression.
Wildtype should establish baseline expression levels.
Wildtype + Vitamin D should result in decreased IL2 levels, thus increased itm2bb levels.
Other immune system gene expression levels will be measured (such as other cytokines, growth factors, MHC class 1 genes) if their homolog exists. This will provide immense insight into understanding how the zebrafish immune system works. The zebrafish immune system can be compared to the human immune system so that differences can be established. These differences can be studied to see why scaled organisms don't get psoriasis.
Future Experiments
After establishing the IL2/itm2bb relationship in zebrafish, the cellular localization of these proteins would be useful for understand their function in apoptosis. Immunostaining would be the preferred method since the antibodies for IL2 and itm2bb have already been made. Also, the tissue localization of these proteins in zebrafish should be determined by examining where GFP is expressed in various tissue samples.
After the immuno-genes' relationships are established, a zebrafish immune system map should created. This zebrafish immuno-pathway can be compared to the human immuno-pathway so that differences can be analyzed. These differences can be studied to see why scaled organisms don't get psoriasis.
After the immuno-genes' relationships are established, a zebrafish immune system map should created. This zebrafish immuno-pathway can be compared to the human immuno-pathway so that differences can be analyzed. These differences can be studied to see why scaled organisms don't get psoriasis.
References
1. Levine N M.D. (2010) "Understanding Psoriasis--the basics." WebMD.
2. Bhalerao J, Bowcock A. The genetics of psoriasis: a complex disorder of the skin and immune system. Hum Mol Genet. 1998;7:1537–1545.
3. Nograles KE, et al. New insights into the pathogenesis and genetics of psoriatic arthritis. Nat Clin Pract Rheum. 2009;5:83–91.
4. Rongioletti F, et al. Psoriasis induced or aggravated by drugs. The Journal of Rheumatology. 2009;83:59–61.
5. Esposito M, et al. An Italian study on psoriasis and depression. Dermatology. 2006. 212:126.
6. Armstrong A M.D. A review of health outcomes in patients with psoriasis. Dermatologic Clinics. 2012;30:1
7. Tiilikainen A, et al. Psoriasis and HLA-Cw6. Br J Dermatol. 1980;102:179–184.
8. Rajan N, et al. Sequence and haplotype analysis supports HLA-C as the psoriasis susceptibility 1 gene. Am J Hum Genet. 2006;78(5):827-51.
9. Bowcock, A and Krueger, J. Getting under the skin: the immunogenetics of psoriasis. Nature Reviews Immunology. 2005;5:699-711
10. STRING
11. MUSCLE
12. FASTA
13. Pfam
14. GO Term Finder
15. Genestier L, Paillot R, Bonnefoy-Berard N, Waldmann H, Revillard JP. T cell sensitivity to HLA class I-mediated apoptosis is dependent on interleukin-2 and interleukin-4. Eur J Immunol. 1997 Feb;27(2):495-9.
16. Bemiss CJ, Mahon BD, Henry A, Weaver V, Cantorna MT Interleukin-2 is one of the targets of 1,25-dihydroxyvitamin D3 in the immune system. Arch Biochem Biophys. 2002 Jun 15;402(2):249-54.
17. Fleischer A et al. ITM2B regulates apoptosis by inducing loss of mitochondrial membrane potential. European Journal of Immunology 32(12): 3498-3505 (2002).
2. Bhalerao J, Bowcock A. The genetics of psoriasis: a complex disorder of the skin and immune system. Hum Mol Genet. 1998;7:1537–1545.
3. Nograles KE, et al. New insights into the pathogenesis and genetics of psoriatic arthritis. Nat Clin Pract Rheum. 2009;5:83–91.
4. Rongioletti F, et al. Psoriasis induced or aggravated by drugs. The Journal of Rheumatology. 2009;83:59–61.
5. Esposito M, et al. An Italian study on psoriasis and depression. Dermatology. 2006. 212:126.
6. Armstrong A M.D. A review of health outcomes in patients with psoriasis. Dermatologic Clinics. 2012;30:1
7. Tiilikainen A, et al. Psoriasis and HLA-Cw6. Br J Dermatol. 1980;102:179–184.
8. Rajan N, et al. Sequence and haplotype analysis supports HLA-C as the psoriasis susceptibility 1 gene. Am J Hum Genet. 2006;78(5):827-51.
9. Bowcock, A and Krueger, J. Getting under the skin: the immunogenetics of psoriasis. Nature Reviews Immunology. 2005;5:699-711
10. STRING
11. MUSCLE
12. FASTA
13. Pfam
14. GO Term Finder
15. Genestier L, Paillot R, Bonnefoy-Berard N, Waldmann H, Revillard JP. T cell sensitivity to HLA class I-mediated apoptosis is dependent on interleukin-2 and interleukin-4. Eur J Immunol. 1997 Feb;27(2):495-9.
16. Bemiss CJ, Mahon BD, Henry A, Weaver V, Cantorna MT Interleukin-2 is one of the targets of 1,25-dihydroxyvitamin D3 in the immune system. Arch Biochem Biophys. 2002 Jun 15;402(2):249-54.
17. Fleischer A et al. ITM2B regulates apoptosis by inducing loss of mitochondrial membrane potential. European Journal of Immunology 32(12): 3498-3505 (2002).
_Site created by Valeri Lapacek
Genetics 677 Assignment, Spring 2012
University of Wisconsin-Madison
Last Updated: 5/23/2012
Genetics 677 Assignment, Spring 2012
University of Wisconsin-Madison
Last Updated: 5/23/2012